- Home
- »
- Medical Devices
- »
-
Leadless Pacemakers Market Size & Share Report, 2030GVR Report cover
![Leadless Pacemakers Market Size, Share & Trends Report]()
Leadless Pacemakers Market Size, Share & Trends Analysis Report By Pacing Chamber (Single Chamber, Dual Chamber), By End-use (Hospitals, Outpatient Facilities), By Region, And Segment Forecasts, 2023 - 2030
- Report ID: GVR-4-68040-138-6
- Number of Pages: 100
- Format: Electronic (PDF)
- Historical Range: 2018 - 2021
- Industry: Healthcare
Leadless Pacemakers Market Size & Trends
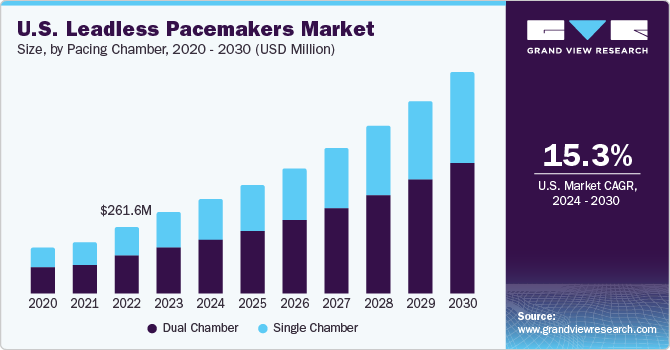
The global leadless pacemakers market size was estimated at USD 514.9 million in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 15.7% from 2023 to 2030. Leadless pacemakers are compact, implantable devices that autonomously manage heart rhythms without conventional wire connections. The market is expected to grow due to several factors, such as the growing geriatric population susceptible to cardiovascular diseases, increasing incidence of bradyarrhythmia, growing product approvals in developed countries, lucrative product pipeline for leadless pacemakers, and its benefits over the traditional pacemakers. According to the Cleveland Clinic., bradycardia symptoms are prevalent in 1 out of every 600 adults aged over 65.
The COVID-19 pandemic moderately impacted the market. Global supply chain disruptions as well as delayed medical procedures in the initial phase hindered industry growth. However, the accelerated telehealth service adoption, along with increased investments in healthcare infrastructure, offer several long-term opportunities for market expansion. Moreover, reimbursement serves as a significant driver in the market. For instance, Medtronic substantially boosted the market as the U.S. Centers for Medicare and Medicaid Services (CMS) approved the Micra Transcatheter Pacing System (TPS) coverage.
Furthermore, the market is expected to benefit due to the growing product pipeline. For instance, Boston Scientific is becoming an upcoming competitor in the leadless pacemaker defibrillator system segment. In December 2022, the Cleveland Clinic successfully implanted Boston Scientific's leadless pacemaker defibrillator system in the first two patients globally as part of a clinical trial. Similarly, in July 2021, MicroPort Scientific Corporation announced to develop leadless pacemaker, with an aim to commercialize the product by 2028.
The adoption of leadless pacemakers is driven by the ability to eliminate a range of complications linked with traditional leads and transvenous pacemakers, such as hematoma, pocket infections, lead fracture, and lead dislodgment. A research study led by the Children’s Hospital of Philadelphia (CHOP) in April 2023 found leadless pacemakers to be safe for pediatric use. According to the research, only 16% of the pediatric patients undergoing leadless pacemaker implantation had experienced a minor complication. Furthermore, the leadless pacemaker has cosmetic interest as there is no visible pacemaker pocket or chest incision.
The increasing product demand can be mainly attributed to the growing global prevalence of cardiovascular disorders, such as bradycardia and atrial fibrillation. This is due to several factors such as unhealthy lifestyle patterns, obesity, and diabetes. As per the CDC, 37.3 million people were diagnosed with diabetes in June 2022. The prevalence of diabetes thus becomes a major contributing factor to the onset of heart diseases, ultimately boosting market growth.
Pacing Chamber Insights
Based on pacing chamber, the market is divided into single chamber and dual chamber segments. The single chamber segment dominated the market with a revenue share of 67.40% in 2022. Single-chamber leadless pacemakers suit a broader range of patients suffering from bradycardia or heart block. Their reliability and simplicity make them a viable choice for patients who do not need advanced features offered by dual-chamber devices. This greater applicability aids their market dominance.
The dual chamber segment is projected to advance at the fastest CAGR of 17.2% through 2030. This growth is attributed to their ability to address a broader range of cardiac conditions. The dual-chamber functionality allows for better synchronization of cardiac contractions and improved hemodynamic performance, enhancing patient outcomes. In February 2022, Abbott significantly advanced leadless pacing technology by successfully implanting the world's first patient with its investigational AVEIR dual-chamber leadless pacemaker system.
End-use Insights
Based on End-use, the market is divided into outpatient facilities and hospitals segments. The hospital segment dominated the market with a revenue share of 89.0% in 2022. Hospitals significantly influence market growth by functioning as crucial centers for patient recruitment and engagement in these trials. For instance, in March 2022, Cleveland Clinic achieved the first successful implantation of the dual-chamber leadless pacemaker system in a U.S. patient as a part of a global clinical trial. The research assessed the safety, functionality, and efficiency of Abbott's AVEIR leadless pacemaker system.
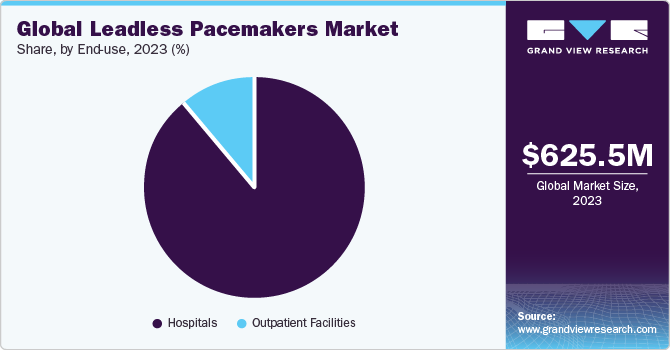
However, the outpatient facilities segment is anticipated to witness the fastest CAGR of 16.5% over the forecast period. The growth can be attributed to technological advancements, accessible training, and improved patient satisfaction. For instance, the European Society of Cardiology has training centers and offers certifications to various outpatient facilities globally that deal with leadless pacemakers to meet the highest standards.
Regional Insights
North America dominated the global market and held 50.71% of the total market share in 2022. The growth can be attributed to technological advancements, increasing heart diseases, and accessibility and availability of skilled professionals. Moreover, in 2020, according to the American Heart Association, Inc., coronary heart disease (CHD) emerged as the primary contributor to cardiovascular disease (CVD)-related fatalities in the U.S., representing 41.2% of such deaths. It emphasizes the imperative for advanced technologies like leadless pacemakers to tackle heart rhythm irregularities linked to CHD and enhance broader cardiovascular health outcomes.
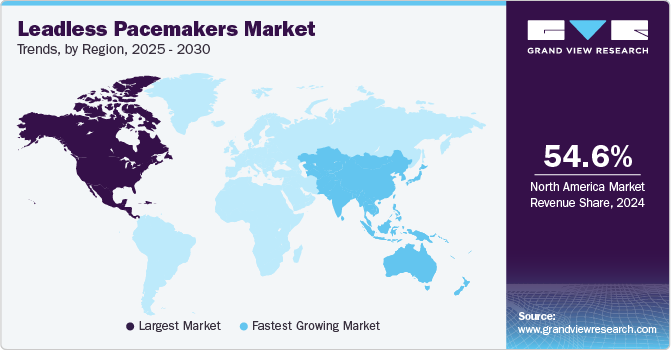
Asia-Pacific is expected to witness the fastest CAGR of 17.0% over the forecast period. The growth can be attributed to the growing collaborations, increasing health expenditure, government initiatives, increasing CVD, and geographic expansion. For instance, in January 2023, Medtronic obtained regulatory approval from the Ministry of Health, Labor, and Welfare in Japan to distribute and reimburse the Micra AV Transcatheter Pacing System (TPS) commercially. Micra AV TPS is the smallest leadless pacemaker to treat AV block patients.
Key Companies & Market Share Insights
The market is less competitive, with a few manufacturers. The market players are focusing on various strategic initiatives such as new product launches, geographical expansion, mergers and acquisitions, collaboration, product upgradation, and partnerships. For instance, in July 2023, the U.S. Food and Drug Administration (FDA) approved Abbott's leadless pacemaker, the AVEIR dual chamber (DR). It is designed to address patients with irregular heart rhythms. Some prominent players in the global leadless pacemakers market include:
-
Abbott
-
Medtronic
Leadless Pacemakers Market Report Scope
Report Attribute
Details
Market size value in 2023
USD 625.5 million
Revenue forecast in 2030
USD 1.74 billion
Growth rate
CAGR of 15.7% from 2023 to 2030
Base year for estimation
2022
Historical data
2018 - 2021
Forecast period
2023 - 2030
Quantitative units
Revenue in USD million/billion and CAGR from 2023 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Pacing chamber, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Germany; UK; France; Italy; Spain; Norway; Denmark; Sweden; China; Japan; India; Australia; Thailand; South Korea; Brazil; Mexico; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Abbott; Medtronic
Customization scope
Free report customization (equivalent up to 8 analyst's working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Leadless Pacemakers Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global leadless pacemakers market report based on pacing chamber, End-use, and region:
-
Pacing Chamber Outlook (Revenue, USD Million, 2018 - 2030)
-
Single Chamber
-
Dual Chamber
-
-
End-use Outlook (Revenue, USD Million, 2018 - 2030)
-
Hospitals
-
Outpatient Facilities
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Sweden
-
Norway
-
Denmark
-
-
Asia Pacific
-
China
-
Japan
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
-
Middle East and Africa
-
Saudi Arabia
-
South Africa
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global leadless pacemakers market size was estimated at USD at USD 514.9 million in 2022 and is expected to reach 625.5 million in 2023.
b. The global leadless pacemakers market is expected to grow at a compound annual growth rate of 15.7% from 2023 to 2030 to reach USD 1.74 billion by 2030.
b. North America dominated the leadless pacemakers market with a share of 50.71% in 2022. This is attributable to technological advancements, increasing heart diseases, the availability of skilled professionals, and the strong presence of industry players in the region.
b. The key players operating in this market are Medtronic and Abbott.
b. Key factors driving the leadless pacemakers market growth are the growing geriatric population susceptible to cardiovascular diseases, increasing incidence of bradyarrhythmia, growing product approvals in developed countries, lucrative product pipeline, and its benefits over the traditional pacemakers
Share this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![ESOMAR Certified Member]()
![Great Place to Work Certified]()
ESOMAR & Great Work to Place Certified
![ISO 9001:2015 & 27001:2022 Certified]()
ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."







